h-index: 18 i10-index: 25
h-index: 18 i10-index: 25
Document Type : Original Research Article
Authors
1 Disaster Risk Management and Development Studies, University of Port-Harcourt, Nigeria
2 Integrated Corrosion Science Co. Ltd., Port-Harcourt, Nigeria
3 University of Port-Harcourt, Nigeria
4 Department Of Geography, University Of Nigeria, University Road, 410001, Nsukka, Enugu State, NigeriaUniversity of Nigeria
5 Institute of Natural Resources, Environment and Sustainable Development, University of Port Harcourt, Nigeria
Keywords
Subjects
Introduction
Oil and gas exploitation and exploration activities can occur onshore, offshore, or in a swamp environment where pipelines are vital in transporting and distributing crude or finished products long distances. Nitonye et al. (2018) [1]. regard pipelines as pipes built to transport liquids, gases, or solid/liquid mixtures over long distances. In the transportation system, pipelines are known as one of the most effective means of transporting goods and services across locations at local, national, and transboundary levels. Such pipelines are built to function for an extended period without failure in their operations; however, they are exposed to various environmental conditions, which they tend to react with, which leads to possible corrosion [2]. Pipelines are -intensive and difficult to access because they are majorly buried deep underground; therefore, ensuring their safety throughout their operational years is essential. Preventive measures must be in place to ensure the pipeline's durability and not compromise its operation, considering the hazards associated with corroded pipelines [3] According to Alawode and Ogunleye (2011) [4], a corroded pipeline coupled with pipeline failure is a potential explosion, human and economic risk, and environmental disaster.
Considering oil and gas activities in the Niger Delta region of Nigeria, Emelu et al. (2021b) [5], noted that corrosion is the third significant cause of oil and gas pipeline destruction aside from sabotage and mechanical failure. The outcome of these pipeline destruction has led to various degrees of environmental degradation [6,7]. human impact [8-14]. and other related impacts [15-18]. Various methods can control corrosion; however, cathodic protection is a significant technique adopted to control the corrosion of pipelines [19].
Globally, pipeline corrosion is one of the major significant challenges faced by the oil and gas industries. This has led to the need for corrosion preventive measures or management when dealing with pipelines as a transportation system in any industry [20]. There are several related oil and gas pipelines corrosion studies [1,20- 27]. however, the present study focused on the disaster-related impact due to inadequate corrosion control through cathodic protection on pipelines in Port Harcourt, Nigeria.
Corrosion
Corrosion, generally, is the dilapidation of a substance from interaction with the environment. A significant quantity of energy is added to metal during its on from ores, making it on from ores and a high-energy condition. The process where metals move to the lower energy oxides is corrosion. The corrosion principle entails the elimination of electrons (oxidation) of the metal (Equation 1-3). In contrast, the electrons are taken up by other atoms that can cause a reduction, such as oxygen or aqueous reduction [28].
![]()
![]()
![]()
The oxidation reaction is popularly known as the anodic reaction, and the reduction reaction is known as the Cathodic Reaction. Corrosion may be categorized into 3 main groups, while corrosion due to microbial activities could be the 4th group (Figure 1). These include categories identified by:
The type and level of pipeline corrosion are determined by various physical and chemical attributes of the environment and the operationality of the pipelines [30,31].
Cathodic Protection (CP) System
According to Guyer (2009)[32], CP minimizes corrosive action by reducing variation in the capacity between the anode and cathode. The reduction is made by applying current from an external source to the pipeline structure. By doing this, the current will potentially ensure that the anode and cathodes are minimized. CP system is generally used to improve the longevity of the potential corrosive structure, and there are two major CP systems: galvanic and impressed current (Figure 2).

Figure 1: a: Pitting corrosion, b: Crevice Corrosion, c: Three-dimensional Reproduction of SCC Shape, d: Uniform Corrosion of Structural Steel. (Sources: [33-35].

Figure 2: Impressed current and galvanic cathodic protection systems
Riemer (2005) [36], states that CP is used to slow down the rate of corroding of a metal surface by making it the cathode of an electrochemical cell (Figure 3). Based on those above, it is known that the amount of corrosion could be limited if all the parts of exposed metal on the surface of a pipeline can be brought under the influence of the current [28].

Figure 3: Principle of Cathodic Protection Systems (Source: Riemer, 2005 [33])
Materials and Method
Study Area
The study was undertaken within Port Harcourt city. The city is located on Latitudes 40 45” N and 40 55” N and Longitudes 60 55” E and 70 05” E. Port Harcourt is approximately 25 km from the Atlantic Ocean, and it is located between the Bonny River and Amadi Creek [37,38]. The central City of Port Harcourt is the Port-Harcourt Local Government Area (LGA) and is the Headquarters of Rivers State [39]. Port-Harcourt metropolis comprises two LGAs (Port Harcourt L.G.A and ObioAkpor LGA) (Figure 4). The study area is found within the tropical monsoon climate, which is characterized by two distinctive seasons; that is the wet season and the dry season. The area is known for low-lying relief with an average elevation of 12 m abl. Generally, the weather and climate attributes of the area have the potential to impact the rate of corrosion of pipeline structures in the area. Considering the soil morphology, Port Harcourt comprises Deltaic plain soil, which is sandy at the surface with little clay content; however, the Clay content increases along the soil profile.

Figure 4: Overview of Port Harcourt Metropolis
Sample Size
The study engaged staff of various oil and gas multinational companies in the Port Harcourt metropolis. The sample size was estimated using the Cochrane formula [40] in Equation (1):
 (1)
(1)
Where;
N= Sample Size
Z= Standard normal deviate correspondingly to the level of significance
p= Prevalence of the study population (p =0.90) from a similar study conducted by Emelu et al., (2021a) [3].
q = 1-p
e = Minimum error @95% confidence interval
Given that e= 0.05, p= 0.50, z= 1.96, q= 1-0.90 =0.5
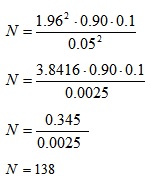
For non-response increase by 10% = 138 + 14 =152
Therefore, a sample size of 150 (staff of Multinationals Oil Companies-MNOCs) was used for the study. Purposive sampling was adopted in selecting 150 respondents for the study, while a structured questionnaire was adopted in data gathering among the respondents.
Data Analysis
Among the 150 questionnaires administered, 100 were filled correctly and fit for further analysis, representing 67%. The retrieved questionnaire was coded on MS Excel and later moved to the Data entry environment of the Statistical Package for Social Sciences (SPSS 22) for proper analysis. As a statistical tool, the descriptive tool consisting of frequency count, percentages, the mean, and standard deviation was adopted for the analysis.
Result and Discussion
The estimated data indicated that 69% of the respondents were site workers, while 31% were management staff. The study noted that all respondents witnessed pipeline leakages at some point in the profession; however, 28% attributed leakage to cathodic rupture, 20% to corrosion, 19% to vandalization, 9% to rust, 2% to age, and wear. In comparison, 22% of pipeline leakage was attributed to corrosion and vandalism, as presented in Figure 5. The finding showed similarity to the study conducted by Steine (2010)[41], Ambituum et al. (2015)[42], and Shittu et al. (2016)[43], which noted corrosion, poor infrastructure, inadequate maintenance, and vandalism, were some of the predominant causes of pipeline leakages. Considering the associated risk due to leakage as a result of inadequate corrosion control, the outcome showed that leakages have environmental risk (91%), production risk (94%), Facility risk (70%), financial risk (85%), and personnel risk (60%) as presented in Figure 5. The outcome noted that the respondents highly rated the risks associated with production and environmental impact. Ebike et al. (2014)[44], noted that the aftermath of an oil spill includes cost implications, a hazard to workers, and environmental pollution. On environmental pollution, Adekola et al. (2017)[10], noted that over 546 million gallons of oil spread across the Niger Delta environment leading to a chronic state of pollution. As a preventive measure, the respondents indicated that the MNOCs carry out maintenance practices on their CPS, and such practices maintain the pipelines.
In contrast, potential soil readings are carried out annually. Respondents indicated that CPS prevents disasters associated with pipeline leakage due to the corrosion effect, and a coating defect survey is carried out annually. Unueroh et al. (2016)[20], asserted that the application of the CP system for arresting corrosion in the pipeline in the oil and gas industry is more effective and provides efficient utilization than other available methods.


Figure 5: Details on variables related to associated risk with inadequate corrosion
Conclusion
It is vital to achieve social and economic gain from the commercialization of oil and gas resources for the development and growth of the producing state or nation; however, there is a more pressing desire to preserve the environment, facility, and staff and certify sustainable production. The CPS is vital in ensuring that all associated disaster risks surrounding pipeline leakage and corrosion can be overcome in the oil and gas industry with little or no environmental impact from the system. Installation of CPS serves as preventive/preparedness and mitigation measures against disaster. Most importantly, it helps to prolong the life span of underground pipelines in the oil and gas industry.
Citation V.O. Emelu, C. Emelu, B.B. Babatunde, E. Wali, O.O. Afolabi*, Associated Disaster Risk of Inadequate Corrosion Control (Cathodic Protection) on Pipelines in Port Harcourt, Nigeria: A Quantitative Approach. J. Eng. Indu. Res. 2023; 4(1):22-30
https://doi.org/10.22034/jeires.2023.1.3
Copyright © 2023 by SPC (Sami Publishing Company) + is an open access article distributed under the Creative Commons Attribution License(CC BY) license (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
| Article View | 188,296 |
| PDF Download | 87,678 |